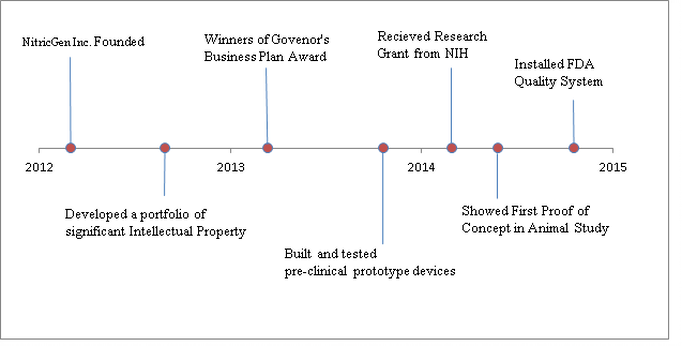
- Continue R&D and get device approval for use on humans.
- Obtain IRB & FDA agreement for a pilot and a clinical trial.
- Complete the clinical trial in chronic diabetic foot ulcers and gain FDA device approval for diabetic foot ulcer therapy.
- Obtain reimbursement codes for insurance and for Medicare/Medicaid.
- Implement a sales force, distribution network, and contract manufacturer.
- Market and sell for the treatment of diabetic foot ulcers.